The global medical rehabilitation robotics market is anticipated to experience rapid growth, with a projected compound annual growth rate (CAGR) of 15.2% between 2023 and 2033. As a result, the market is expected to reach a valuation of approximately US$ 1.76 Billion by the end of 2033, representing a significant increase from its current valuation of US$ 366.2 Million in 2022. This expansion can be attributed to various factors, including advancements in robotic technology, growing demand for minimally invasive procedures, and increasing awareness regarding the benefits of medical rehabilitation robotics.
The WHO states that the year 2021 witnessed 1.71 Billion people suffering from musculoskeletal disorders. The figure certainly calls for utilization of rehabilitation robots as the devices do aid physically challenged or differently abled patients in balancing, standing up, and maintaining gait.
Medical rehabilitation robotics have innumerable benefits – like they do offer an opportunity to that contracting spinal cord injury to stand and walk at a lower metabolic cost. The market is also home to several technological advancements, like incorporating ML and AI with several advanced sensors. Exploding geriatric population is another factor contributing to the growth of medical rehabilitation robotics market.
Claim your complimentary report with captivating visuals.
https://www.futuremarketinsights.com/reports/sample/rep-gb-14242
Along the above-mentioned lines, PARO comes across as companion robot that has been specially designed for those suffering from Alzheimer’s, dementia, and various other cognition disorders. Dr. Takanori Shibata from Japan’s National Institute of Advanced Industrial Science and Technology has come up with this assistive robot for helping those with disabilities fulfil daily activities like eating and walking.
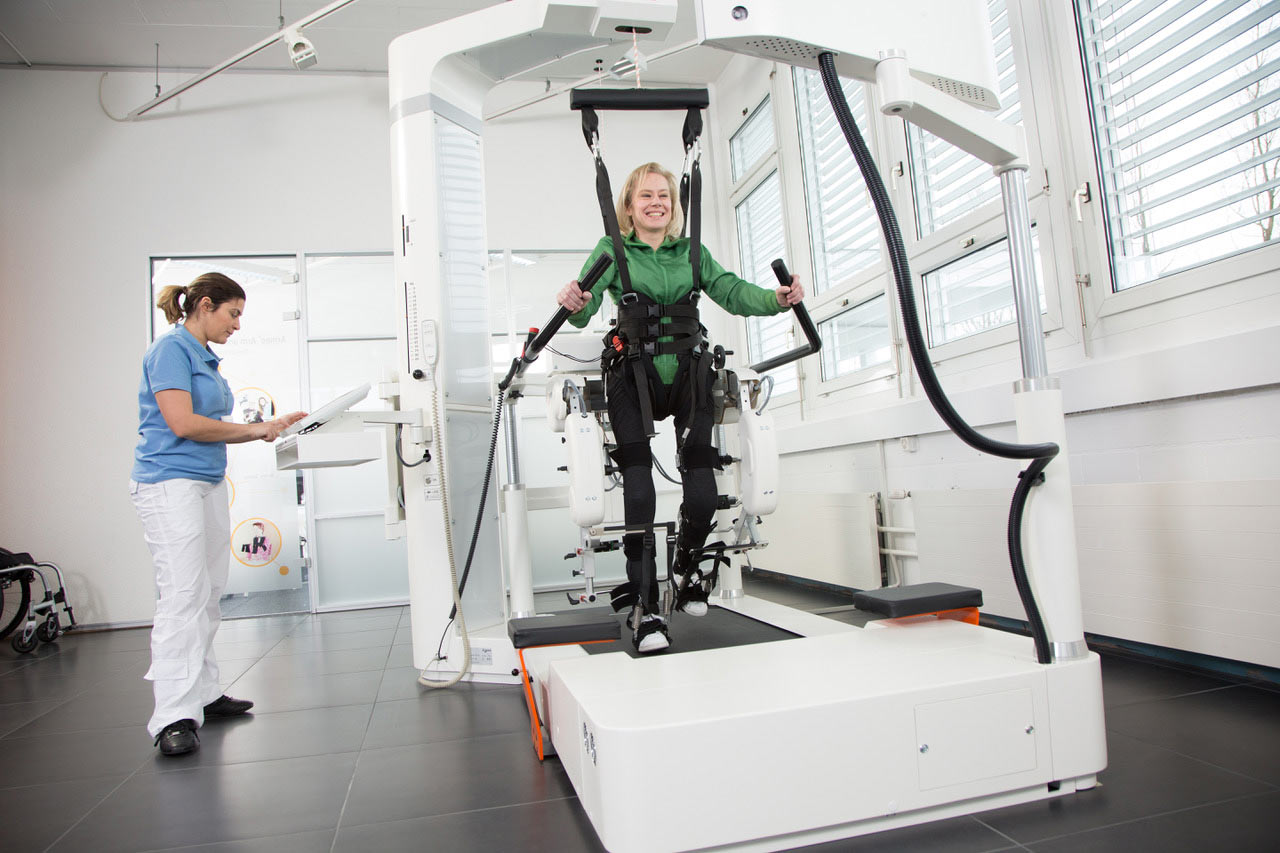
Robotic lower-body exoskeleton is being used to assist the ones with paralysis of the lower limbs and does encourage muscle activity therein.
Future Market Insights has walked through these facts with future perspectives in its latest market study entitled ‘Medical Rehabilitation Robotics Market’ with its team of analysts and consultants deploying a 360-degree view in its primary, secondary, and tertiary modes of research.
“With Spike in cases of stroke, the global medical rehabilitation robotics market is expected to go great guns in the near future”, says an analyst from Future Market Insights.
Key Takeaways from Medical Rehabilitation Robotics Market
- North America holds more than 40% of the market share due to growing incidences of spinal cord injury followed by rising aging and disabled population.
- Europe holds a significant market share with Germany leading from the front with Spain, Denmark, and the UK leading from the front.
- The Asia-Pacific is expected to grow at a rapid pace in the medical rehabilitation robotics market with countries like Japan obtaining the status of ‘super-aging society’.
Understanding the study’s underlying assumptions. Ask an Analyst Now
https://www.futuremarketinsights.com/ask-question/rep-gb-14242
Competitive Robotics
- Ekso Bionics Holdings, Inc. received approval from the US FDA for marketing the EksoNR robotic exoskeleton for usage with patients suffering from MS (Multiple Sclerosis).
- Tyromotion, of late, completed acquisition of yband therapy AG (a Swiss-based start-up) for encouraging patients’ ownership of recovery with cutting-edge movement trackers.
- Bionik Laboratories Corporation, in January 2019, did launch InMotion ARM/HAND robotic system to clinically rehabilitate stroke survivors and the ones having mobility impairments owing to neurological conditions. The enhanced robotic system has been developed according to principles of motor learning as well as neuro-plasticity and makes use of AI and data analysis for offering individualized therapy to the patients.
- ReWalk Robotics, Inc., in June 2019, did announce that the US FDA approved the ReStore soft exo-suit system for selling to the rehabilitation centers all over the US.
- Cyberdyne Inc., in January 2018, obtained approval from the US FDA to sell HAL (Hybrid Assistive Limb) lower-body exoskeleton in the licensed medical facilities all over the US.
- Stryker Corporation, in the year 2020, did receive regulatory approval for MAKO robotic surgery system in Japan for partial knee indication.
- Accuray, in 2020, did launch CyberKnife S7 System, which comes across as a next-generation CyberKnife platform with real-time, advanced, AI-driven motion tracking as well as synchronization treatment delivery for SBRT (stereotactic body radiation therapy) and SRS (stereotactic radiosurgery).
- Medtronic, in 2020, completed acquisition of Medicrea through AI, patient specific implants, and predictive modeling.
- Intuitive Surgical, in 2019, did receive clearance from the US FDA regarding da Vinci Single-port Surgical System.
Key Market Players:
- Ekso Bionics Holdings Inc.
- AlterG Inc.
- Bionik Laboratories Corp.
- Cyberdyne Inc.
- Rewalk Robotics.
- Instead Technoligies Ltd.
- Hocoma AG.
- Fourier Intelligene
- Tyromotion Gmbh
- Toyota Motor Corporation
- BeatBots LLC
- Bioxtreme
- Rex Bionics Inc.
- LEADERS REHAB ROBOT Co. Ltd.
Access Exclusive Market Insights – Purchase Now!
https://www.futuremarketinsights.com/checkout/14242
Key Segments Covered in Medical Rehabilitation Robotics Industry Research
By Product:
- Exoskeletons
- Therapeutic/ Assistive
By Extremity:
- Upper Extremity
- Arm
- Wrist
- Shoulder
- Elbow
- Lower Extremity
By Therapy:
- Limb Mobility
- Gait
- Sensory
By Patient:
- Adults
- Pediatric
By Application:
- Neurorehabilitation
- Physical Rehabilitation
- Others
By End user:
- Hospitals
- Wellness centers
- Rehabilitation centers
- Homecare
About Future Market Insights (FMI)
Future Market Insights, Inc. (ESOMAR certified, recipient of the Stevie Award, and a member of the Greater New York Chamber of Commerce) offers profound insights into the driving factors that are boosting demand in the market. FMI stands as the leading global provider of market intelligence, advisory services, consulting, and events for the Packaging, Food and Beverage, Consumer Technology, Healthcare, Industrial, and Chemicals markets. With a vast team of over 5,000 analysts worldwide, FMI provides global, regional, and local expertise on diverse domains and industry trends across more than 110 countries.
Contact Us:
Future Market Insights Inc.
Christiana Corporate, 200 Continental Drive,
Suite 401, Newark, Delaware – 19713, USA
T: +1-845-579-5705
For Sales Enquiries: sales@futuremarketinsights.com
Website: https://www.futuremarketinsights.com
LinkedIn| Twitter| Blogs | YouTube